Instead of downgrading to an older, insecure version of Firefox and changing your update settings, you should consider these alternatives: Install Firefox Extended Support Release. Firefox Extended Support Release (ESR) is an official version of Firefox developed for large organizations like universities and businesses. Firefox ESR does not. (b) A local law enforcement authority may not provide notice to the superintendent or any administrator under Article 62.053(e) or 62.055(f) if the basis on which the person is subject to registration is a conviction, a deferred adjudication, or an adjudication of delinquent conduct for an offense under Section 25.02, Penal Code, or an offense. In another study, when a lower dose of 200 mg of HCQ twice daily was added to basic treatment, mortality of even critically ill patients was significantly reduced. These and our findings indicate that proper dosing of HCQ with its long half-life might be key for a favourable outcome of COVID-19 patients. Download MusConv - Version 4.9.63 MusConv moves playlists, albums, followed artists and liked songs across various services – Spotify, Apple Music, Google Music, Amazon Music, YouTube, YouTube Music, iTunes, Deezer, Tidal, SoundCloud and others! Transfer your playlists, albums, followed artists and liked tracks between 50+ music services.
The [OH-] of an aqueous solution is #4.62 * 10^-4# M. What is the pH of this solution?
1 Answer
Explanation:
For pure water at #25^@'C'#, the concentration of hydronium ions, #'H'_3'O'^(+)#, is equal to the concentration of hydroxide ions, #'OH'^(-)#.
More specifically, water undergoes a self-ionization reaction that results in the formation of equal concentrations of hydronium and hydroxide anions.
#2'H'_2'O'_text((l]) rightleftharpoons 'H'_3'O'_text((aq])^(+) + 'OH'_text((aq])^(-)#
At room temperature, the self-ionization constant of water is equal to
#K_W = ['H'_3'O'^(+)] * ['OH'^(-)] = 10^(-14)#
This means that neutral water at this temperature will have
#['H'_3'O'^(+)] = ['OH'^(-)] = 10^(-7)'M'#
As you know, pH and pOH are defined as
#'pH' = - log( ['H'_3'O'^(+)])#
Office 365 monthly uk. #'pOH' = - log(['OH'^(-)])#
and have the following relationship

#'pH' + 'pOH' = 14#
In your case, the concentration of hydroxide ions is bigger than #10^(-7)'M'#, which tells you that you're dealing with a basic solution and that you can expect the pH of the water to be higher than #7#.
A pH equal to #7# is characteristic of a neutral aqueous solution at room temperature.
So, you can use the given concentration of hydroxide ions to determine the pOH of the solution first
Another Version 625
#'pOH' = - log( 4.62 * 10^(-4)) = 3.34#
This means that the solution's pH will be
#'pH' = 14 - 'pOH'#
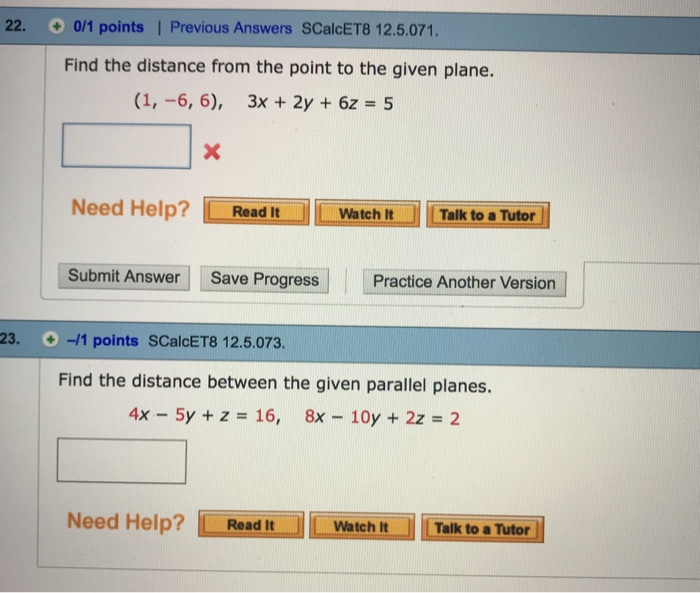
#'pH' + 'pOH' = 14#
In your case, the concentration of hydroxide ions is bigger than #10^(-7)'M'#, which tells you that you're dealing with a basic solution and that you can expect the pH of the water to be higher than #7#.
A pH equal to #7# is characteristic of a neutral aqueous solution at room temperature.
So, you can use the given concentration of hydroxide ions to determine the pOH of the solution first
Another Version 625
#'pOH' = - log( 4.62 * 10^(-4)) = 3.34#
This means that the solution's pH will be
#'pH' = 14 - 'pOH'#
#'pH' = 14 - 3.34 = color(green)(10.66)#
Indeed, the pH is higher than #7#, which confirms that you're dealing with a basic solution.
Related questions
| Printable version |
Another Version 33
|
Another Version 20
|

